Search Thermo Fisher Scientific
Thermo Scientific Chemicals
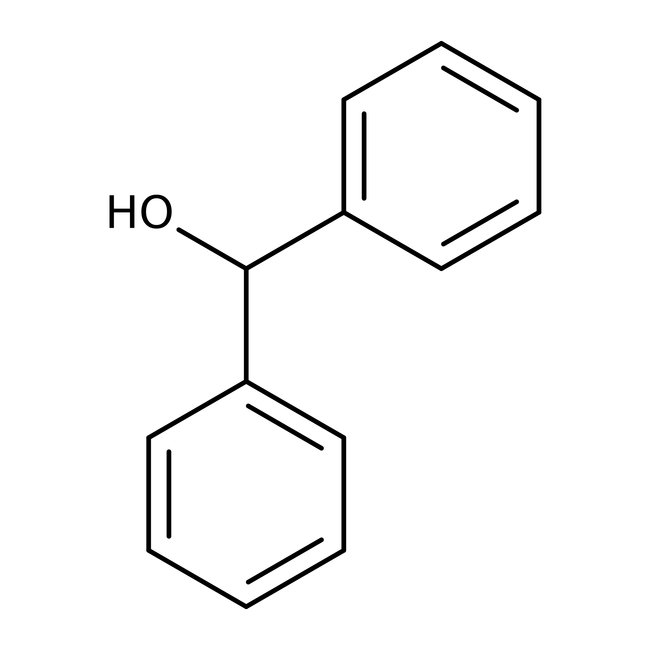
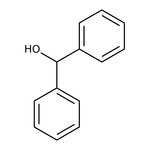
Benzhydrol, 99%, Thermo Scientific Chemicals
CAS: 91-01-0 | C13H12O | 184.238 g/mol
| Catalog Number | Quantity |
|---|---|
| ALFA12884.30 | 250 g |
Catalog number ALFA12884.30
View Price:Sign InSign in to see your account pricing. Need an account? Register with us today.
Quantity:
250 g
Specifications
Chemical Name or MaterialBenzhydrol
CAS91-01-0
Health Hazard 1H315-H319-H335
Health Hazard 2GHS H Statement
H315-H319-H335
Causes skin irritation.
Causes serious eye irritation.
May cause respiratory irritation.
H315-H319-H335
Causes skin irritation.
Causes serious eye irritation.
May cause respiratory irritation.
Health Hazard 3P261-P264b-P271-P280-P302+P352-P304+P340-P305+P351+P338-P312-P332+P313-P362-P501c
View more
Benzhydrol is widely used as intermediates in pharmaceuticals (including antihistamines), agrochemicals, perfumes and other organic compounds. It is used as a fixative in the perfume industry. It is involved in polymerization reaction as a terminating group. It is used as precursor to prepare modafinil, benztropine and diphehydramine.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Benzhydrol is widely used as intermediates in pharmaceuticals (including antihistamines), agrochemicals, perfumes and other organic compounds. It is used as a fixative in the perfume industry. It is involved in polymerization reaction as a terminating group. It is used as precursor to prepare modafinil, benztropine and diphehydramine.
Solubility
Slightly soluble in water.
Notes
Incompatible with strong oxidizing agents, acids, acid chlorides and acid anhydrides.
Benzhydrol is widely used as intermediates in pharmaceuticals (including antihistamines), agrochemicals, perfumes and other organic compounds. It is used as a fixative in the perfume industry. It is involved in polymerization reaction as a terminating group. It is used as precursor to prepare modafinil, benztropine and diphehydramine.
Solubility
Slightly soluble in water.
Notes
Incompatible with strong oxidizing agents, acids, acid chlorides and acid anhydrides.
RUO – Research Use Only
General References:
- The protection of OH groups as benzhydryl ethers can be catalyzed by sulfuric acid: Org. Synth. Coll., 4, 72 (1963); Angew. Chem. Int. Ed., 15, 281 (1976). See also Benzhydryl bromide, L02211. Thiols, e.g. in cysteine residues in peptide synthesis, can be protected as thioethers in the presence of TFA or HBr/AcOH: J. Chem. Soc. (C), 2683 (1970). The group can be cleaved by TFA in the presence of a cation scavenger, e.g. phenol or anisole (same ref.), or by Na in liquid NH3: J. Am. Chem. Soc., 84, 3887 (1962). See Appendix 6.
- Carboxyl groups can be protected by esterification in benzene with tosic acid catalyst. Deprotection can be effected with the same catalyst by refluxing in toluene, which undergoes electrophilic alkylation: Tetrahedron Lett., 37, 1965 (1996).
- Misawa, T.; Dodo, K.; Ishikawa, M.; Hashimoto, Y.; Sagawa, M.; Kizaki, M.; Aoyama, H. Structure-activity relationships of benzhydrol derivatives based on 1'-acetoxychavicol acetate (ACA) and their inhibitory activities on multiple myeloma cell growth via inactivation of the NF-κB pathway. Bioorg. Med. Chem. 2015, 23 (9), 2241-2246.
- Taskin, O. S.; Temel, B. A.; Tasdelen, M. A.; Yagci, Y. Synthesis of block copolymers by selective H-abstraction and radical coupling reactions using benzophenone/benzhydrol photoinitiating system. Eur. Polym. J. 2015, 62, 304-311.